Natural Parasite Control for Horses: Beyond the Chemical Rotation
- Roots Mercantile

- 5 hours ago
- 15 min read
Why blanket deworming is accelerating resistance — and how a terrain-based approach builds the kind of host immunity that keeps worm burdens low from the inside out.
By Le Anna K. | Rooted Saviors | Biofield App | Stewards Under Pressure
Ask most horse owners about their deworming protocol and you'll hear a familiar answer: every six to eight weeks, rotating between drug classes, treating every horse on the property at the same time. This approach has been standard practice for decades. It is also, according to the latest veterinary parasitology research, accelerating one of the most serious problems in equine health: widespread drug resistance in the worm populations that live inside our horses.
This post isn't about abandoning deworming — there are horses that genuinely need chemical treatment, and there are situations where it is the right and necessary choice. This post is about understanding why the blanket rotation model is failing, what the science says about a smarter approach, and — perhaps most importantly — what building the horse's own terrain-based resistance actually looks like in practice.
Because here is the truth that the rotation model misses entirely: horses are not passive hosts. Their immune system, gut health, and mineral status have a profound effect on how many worms establish, how many eggs are shed, and how rapidly a population can be controlled without chemical intervention.
"The goal is not to eliminate all worms — it is to keep the horse healthy despite the presence of worms. That distinction changes everything about how we approach parasite management."
Why the Rotation Model Is Creating Resistant Worms
The logic behind rotating between dewormers seemed sound when it was introduced in the 1960s: use drugs from different chemical classes to prevent any one drug becoming ineffective. Rotate often enough and worms won't have time to build resistance to any particular compound.
The problem is that this logic has a fatal flaw — and it took decades of practice to make it visible. By treating every horse on a schedule regardless of their actual worm burden, blanket rotation does something counterproductive: it removes the susceptible worm population (the ones that can be killed by the drug) and leaves only the resistant survivors. Those survivors then breed the next generation. With each treatment cycle, the proportion of resistant worms in the population grows.

Figure 1: The chemical rotation resistance cycle — how blanket deworming progressively builds resistant worm populations while damaging gut terrain.
The 2021 World Association for the Advancement of Veterinary Parasitology (WAAVP) guidelines for equine parasite control explicitly state that targeted selective treatment — treating only horses with high egg counts rather than all horses on a schedule — is now the recommended approach. Studies across Europe and North America have found anthelmintic resistance in cyathostomes (small strongyles) to be widespread, with some farms showing resistance to two of the three major drug classes used in equine deworming.
There is a second problem with blanket chemical rotation that the terrain model makes visible: every deworming treatment disrupts the gut microbiome. Broad-spectrum anthelmintics affect the intestinal environment in ways that reduce microbial diversity and damage the mucosal lining — the same lining that houses 80% of the immune cells responsible for regulating the horse's natural response to parasites. Treating a horse chemically every six weeks means the gut terrain is continuously disrupted before it can fully recover — creating the very conditions of immune suppression and mucosal weakness that make the horse more vulnerable to heavy worm burdens.
Host Resistance — The Part We Rarely Talk About
One of the most consistent findings in equine parasitology research is that within any group of horses on the same pasture, a small proportion of horses account for the vast majority of egg shedding. Studies repeatedly show that roughly 20% of horses shed approximately 80% of the parasite eggs in a population. The other horses — same environment, same grazing — maintain low or negligible egg counts.
This is not random. It reflects something real about the internal state of those horses — what parasitologists call host resistance. And host resistance is, to a significant degree, a terrain variable.

Figure 2: Host resistance — how the horse's internal terrain determines worm burden from the same pasture exposure.
A horse with strong host resistance is not necessarily a horse with no worms. All grazing horses ingest larvae. What differs is what happens after ingestion: how many larvae successfully establish in the gut wall, how many mature to egg-laying adults, and how many eggs are shed back onto pasture to reinfect. A horse with robust immune function, intact gut mucosal barriers, and low oxidative stress is able to limit establishment and egg shedding through active immune mechanisms — including IgA antibody responses in the gut wall and mast cell activity that disrupts larval development.
These immune mechanisms require resources: minerals to power immune cell function, an intact mucosal lining to deploy those cells effectively, and low enough baseline oxidative stress that the immune system is not already overwhelmed by other demands. This is why terrain work — remineralizing, gut healing, reducing oxidative load — is not a soft adjunct to parasite control. It is the mechanism through which the horse's own body becomes a less hospitable environment for parasites.
The Main Equine Parasites — A Plain-Language Overview
Before building a management approach, it helps to understand which parasites matter most and why:
Small strongyles (cyathostomes) — the primary concern in adult horses
These are the most significant parasitic threat in horses over the age of three. Up to 50 species of small strongyles can infect horses, and their most dangerous capacity is encysting — burrowing into the gut wall and remaining dormant for months or years in a larval stage that no current drug can reliably reach. When large numbers of encysted larvae emerge simultaneously (a condition called larval cyathostominosis), the result can be severe colitis, rapid protein loss, and life-threatening diarrhea. Fecal egg counts monitor the adult population but do not reveal the encysted burden — making clinical observation of the horse's condition equally important.
Large strongyles (Strongylus vulgaris) — serious but controlled
Historically the most dangerous equine parasite, Strongylus vulgaris migrates through arterial walls and can cause colic and serious vascular damage. Modern deworming programs have dramatically reduced its prevalence, and it is now uncommon in well-managed horse populations. It remains worth testing for, as it is serious when present.
Tapeworms (Anoplocephala perfoliata)
Tapeworms require a mite intermediate host and are concentrated near the ileocecal junction, where heavy burdens have been associated with a specific type of colic (spasmodic and ileocecal intussusception). Routine fecal egg count tests do not detect tapeworms — a saliva-based ELISA test or tapeworm-specific fecal test is needed. Treatment requires praziquantel or a double dose of pyrantel, typically once or twice a year.
Bot flies (Gasterophilus spp.)
Bots are fly larvae, not true worms, and complete their larval development in the horse's stomach. They are irritating but not typically dangerous in small numbers. Management focuses on removing bot eggs from the coat during fly season and treating with ivermectin in late autumn after the first frost kills adult flies.
Ascarids (Parascaris equorum) — the foal parasite
Ascarids are the primary parasitic concern in foals and youngsters under two years old. They are uniquely dangerous because young horses have not yet developed adult immune resistance, and heavy burdens can cause impaction colic and lung damage during larval migration. Ascarid resistance to ivermectin and moxidectin is now well-documented — fenbendazole or pyrantel are the appropriate treatment choices for foals.
Start with a Fecal Egg Count — Know Before You Treat
The single most important shift you can make in your parasite management approach costs very little and takes the guesswork out of every decision that follows: test before you treat.
A fecal egg count (FEC) measures the number of worm eggs per gram of manure (epg). It gives you real information about each individual horse's worm burden rather than assuming all horses are equal. This matters because they are not — and treating a horse with a low burden contributes to resistance without any meaningful benefit to that horse.
General FEC interpretation guidelines (these vary slightly by source — always discuss with your vet):
• Under 200 epg — low shedder: no chemical treatment indicated; terrain support and retest in 3 months
• 200–500 epg — moderate shedder: terrain work, herbal support, retest in 6–8 weeks; discuss with vet whether treatment is needed
• Over 500 epg — high shedder: targeted chemical treatment indicated alongside terrain work
Research consistently shows that in any managed herd, 50–70% of horses will be low shedders who do not need chemical deworming at any given time. Identifying and not treating these horses preserves refugia — a population of drug-susceptible worms on pasture that dilutes the resistant population and slows the development of full resistance.
After treating high shedders, a fecal egg count reduction test (FECRT) — resampling 10–14 days after treatment — tells you whether the drug you used was effective. Egg count reductions below 95% for benzimidazoles, 90% for pyrantel, or 99% for ivermectin/moxidectin suggest resistance in the worm population on your property. This information is invaluable for making future decisions.
Building the Terrain — The Four-Layer Approach
Regardless of a horse's FEC result, terrain work runs in the background of every parasite management decision. A horse with strong host resistance maintains lower egg counts over time, recovers faster from any worm burden, and is less likely to show clinical disease even when some parasite load is present. This is the layer of management that the chemical rotation model ignores entirely — and it may be the most important one for long-term parasite health.
Layer 1: Remineralize — power the immune response
Parasite-specific immunity in horses depends on immunoglobulin A (IgA) antibodies in the gut wall, mast cell activity, and eosinophil responses — all of which require adequate mineral status to function. Zinc deficiency specifically impairs the production and activity of T-helper cells, which orchestrate the immune response to gut parasites. Selenium deficiency impairs neutrophil and macrophage activity. Copper and iron are required for the oxidative enzyme systems that immune cells use to destroy invading organisms.
Practical remineralization for parasite resistance:
• Seaweed meal or kelp — broad-spectrum bioavailable trace minerals including iodine, which supports thyroid function and immune coordination
• Free-choice mineral salt with a full trace mineral profile — allow the horse to self-regulate intake according to their own needs
• Magnesium — stabilizes mast cell membranes (mast cells are a key component of the gut's antiparasitic immune response) and regulates the stress hormone cortisol that suppresses immunity
• Zinc and selenium at appropriate levels for your region's soil — have hay or pasture tested where possible, as regional soil deficiencies vary significantly
Layer 2: Gut health — maintain the mucosal immune barrier
The gut wall is the first line of defense against parasite establishment. Larvae that are ingested must penetrate the mucosal lining to encyst or complete their development — a mucosal barrier that is intact, well-supported by beneficial microbiota, and low in inflammation is significantly harder to penetrate than one that is inflamed, thin, or dysbiotic.
Horses on high-starch diets, horses with gastric ulcers, and horses receiving frequent antibiotic or NSAID courses all have compromised mucosal integrity that directly impairs this first line of parasite defense. Restoring it requires:
• Forage-first feeding — high-quality hay available continuously supports hindgut microbial balance and maintains the buffering that protects the mucosal lining from acid damage
• Slippery elm bark — coats and soothes the mucosal lining, reduces gut wall inflammation, provides prebiotic fiber for beneficial bacteria
• Aloe vera juice (inner leaf, without aloin) — mucosal-protective and anti-inflammatory; particularly useful for horses with a history of NSAID use or gastric ulceration
• Fermented feeds or quality equine probiotics — support microbiome diversity, which directly influences the local immune tone of the gut wall
Layer 3: Antiparasitic herbs — reduce the larval load naturally
Several plants produce compounds that are genuinely antiparasitic — not in the sense of replacing a veterinary anthelmintic when one is clinically indicated, but in the sense of creating a gut environment that is less hospitable to larval development, reducing egg shedding, and supporting the horse's own expulsive immune mechanisms. These are supportive tools used alongside terrain work, not standalone treatments.
The most evidence-supported options for equine use include:
• Black seed (Nigella sativa) — has specific research support in equine parasite studies, with one peer-reviewed trial showing significant reduction in egg counts and adult worm numbers in horses treated with black seed compared to controls. Contains thymoquinone, which has demonstrated anthelmintic activity
• Wormwood (Artemisia absinthium) — traditional antiparasitic herb with documented activity against intestinal parasites; contains absinthin and other bitter compounds that create an inhospitable gut environment for larval establishment. Use in rotation and not continuously
• Garlic — contains allicin and other sulfur compounds with antiparasitic and immune-supporting properties; horses generally accept it in feed; use in moderation and not in horses with any hemolytic conditions
• Diatomaceous earth (food grade) — microscopic silica particles that mechanically damage the cuticle of soft-bodied larvae; works physically rather than chemically; particularly useful in feed to reduce gut larval establishment. Not effective against encysted larvae
• Pumpkin seeds — contain cucurbitacin, which paralyses adult worms and aids their expulsion; a traditional remedy with some modern research support; safe and palatable in feed
• Papaya seeds and green papaya — contain papain and carpaine with documented anthelmintic properties; used in traditional equine management in tropical regions
Important note: these herbs and foods are terrain supporters and herd management tools, not replacements for targeted chemical treatment in horses with confirmed high burdens or clinical parasite disease. They work best as part of a sustained daily or weekly protocol that builds the internal environment over months.
Layer 4: Stress reduction and grounding — the overlooked immune inputs
Cortisol — the stress hormone elevated by transport, social disruption, competition, stall confinement, and irregular management — directly suppresses the IgA and mast cell responses that constitute equine gut immunity to parasites. A horse under chronic low-grade stress is immunologically less capable of limiting parasite establishment regardless of what herbs or minerals they receive.
Maximizing turnout, maintaining consistent management routines, providing social contact appropriate to the horse's needs, and minimizing unnecessary transport during high-risk periods (spring and autumn, when larval populations on pasture peak) all contribute to the parasitological health of the horse.
Grounding — direct contact between hooves and natural earth — provides free electrons from the Earth's surface that neutralize the reactive oxygen species driving gut inflammation and oxidative immune suppression. Horses with consistent pasture time on natural ground maintain lower baseline inflammatory states and better gut mucosal integrity than stall-kept counterparts. This is not incidental to parasite management — it is foundational to the immune terrain that determines host resistance.
Pasture Management — Reducing the Environmental Load
The terrain approach works inside the horse. Pasture management works outside — reducing the number of infective larvae the horse encounters in the first place, both are necessary components of an integrated program.
Worm larvae on pasture are killed by heat, drought, and ultraviolet light. They survive best in cool, moist conditions in spring and autumn — which is exactly when grazing pressure tends to be highest. Practical pasture management strategies:
• Rotate grazing between paddocks — allow paddocks to rest for at least six to eight weeks before returning horses to them. Longer rest periods (three months or more) are significantly more effective in warm climates where larval development is faster
• Harrow and chain pastures — dragging the surface breaks up manure pats and exposes larvae to drying and sun. Do this only in hot, dry weather — harrowing in cool, damp conditions can spread larvae rather than killing them
• Remove manure regularly — particularly from small paddocks and sacrifice areas where horses graze intensively. The goal is to reduce the larval bank that accumulates in heavily used areas
• Mixed species grazing — cattle and sheep will graze off equine parasite larvae (which cannot infect them) and their own parasites cannot infect horses. Mixed grazing on the same pasture is one of the most effective larval reduction strategies available
• Avoid overgrazing — horses preferentially avoid grazing near their own manure, but overgrazing removes this luxury and forces contaminated grass consumption
• Be cautious about harrowing in autumn — larvae harrowed in cool, damp autumn conditions may survive and redistribute. In cooler climates, harrowing is most useful in summer.
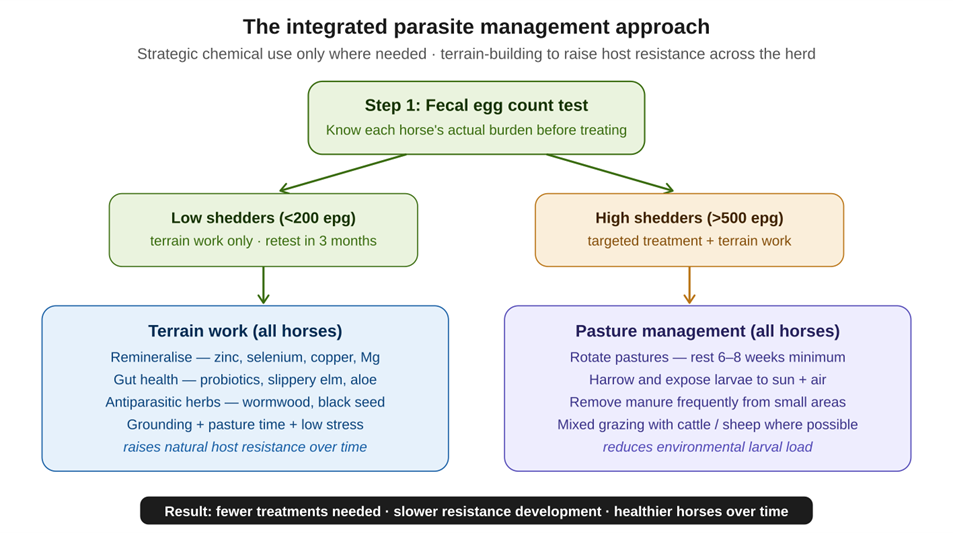
Figure 3: The integrated parasite management approach — fecal egg count testing guides targeted treatment, while terrain work and pasture management reduce the burden from both directions.
A Practical Starting Protocol
If you are new to this approach and transitioning from a blanket rotation schedule, here is a sensible way to begin:
• Spring and autumn: collect a fecal sample from every horse and submit for a standard fecal egg count. Use the results to determine which horses — if any — need treatment at that time
• Treat only horses with counts above 500 epg, or those with clinical signs of parasitism, following your vet's guidance on drug choice based on known resistance patterns in your region
• For all horses regardless of FEC result: begin the terrain protocol — remineralize, support gut health, introduce antiparasitic herbs at appropriate doses, and maximize natural ground time
• Retest high shedders 10–14 days after treatment (FECRT) to assess drug efficacy; retest all horses three months after the initial count to track trends
• Implement one pasture management change per season — even a single rotation improvement makes a measurable difference
• Keep records — FEC results over time tell you which horses are chronic high shedders (the 20% that need the most attention) and how the herd's overall burden trends as terrain work accumulates
This approach takes more intention than writing a reminder on the calendar for every six weeks. But it produces something the rotation model never can: a herd whose internal terrain is actively working to limit parasite burden — alongside a targeted use of the chemical tools that remain effective precisely because they are being conserved.
Special Considerations — Foals, Youngsters, and Pregnant Mares
Foals and youngsters under two
Young horses have not yet developed adult-level immune resistance to parasites. Ascarids are the primary concern in foals, with the added challenge that ivermectin and moxidectin resistance in ascarids is now common and well-documented. Fenbendazole or pyrantel are the drugs of choice for ascarid treatment in foals. Foals should be tested at approximately eight weeks of age and then every one to three months through their first year. The terrain approach still applies — supporting gut health and mineral status in young horses lays the foundation for adult host resistance — but should not replace appropriate targeted treatment in this vulnerable age group.
Pregnant and lactating mares
Periparturient immune suppression — the natural immunological relaxation around foaling that allows the mare to carry a foal to term — also increases susceptibility to parasite burden. Egg shedding typically rises in late pregnancy and early lactation. Testing mares during late pregnancy and treating if indicated (with products safe for use in late gestation — discuss with your vet) reduces the pasture contamination that foals will be born onto. Terrain support is particularly important during this period to moderate the immune suppression and limit unnecessary worm burden increase.
Horses new to a property
Any horse arriving from another property should be quarantined and tested before being introduced to shared pasture. Treat if indicated (using a drug appropriate to the worm population, with your vet's guidance) during the quarantine period to avoid importing resistant worm populations from their previous environment. Introduce terrain support from day one — the stress of transport and relocation is exactly the kind of immune-suppressing event that allows worm burdens to spike.
The Bigger Picture
Parasite management is one of the areas where the terrain model and conventional medicine most clearly need each other. Chemical anthelmintics, used strategically and only when genuinely indicated, remain important tools — particularly for high shedders, for foals, and during the periparturient period. We are not arguing against them.
What we are arguing is that the rotation model — designed for an era before drug resistance was a recognized problem — is no longer adequate as a standalone strategy. The worm populations inside our horses are evolving faster than we can develop new drug classes. And the response to that problem cannot be more chemicals at more frequent intervals.
The response is a smarter approach: test before you treat. Treat only those who need it. And invest — consistently, daily — in the terrain that makes every horse a more capable, more resistant host. A horse whose gut is healthy, whose minerals are adequate, whose stress is managed, and who spends real time on natural ground is a horse that puts natural limits on what parasites can do inside them.
That is the goal. Not a horse with zero worms — that is neither achievable nor, in small numbers, even desirable for immune development. But a horse whose internal environment is strong enough that parasites are kept in their place, and whose immune system meets every spring and autumn larval challenge from a position of genuine readiness.
The horse that controls its own worm burden is not a horse on a perfect chemical schedule. It is a horse with a healthy gut, full minerals, low oxidative stress, and daily contact with the earth. Build that horse — and the parasites will be far less of a problem.
To explore more about terrain-based equine wellness at Rooted Saviors, visit rootedsaviors.com.
Note: This post is for informational purposes. All parasite management decisions — including deworming schedules, drug selection, and dosing — should be made in consultation with a qualified veterinarian. Fecal egg counts and drug resistance testing should be interpreted by a professional familiar with your region's parasite populations.
Sources & Further Reading
1. Nielsen M.K. et al. (2021). WAAVP guideline for equine parasite control — Veterinary Parasitology — international consensus on targeted selective treatment and resistance management.
2. Traversa D. & von Samson-Himmelstjerna G. (2016). Anthelmintic resistance in equine parasites — International Journal for Parasitology: Drugs and Drug Resistance — current state of drug resistance across worm classes.
3. Peregrine A.S. et al. (2014). Anthelmintic resistance in equine parasites — a multinational survey — Equine Veterinary Journal — prevalence of resistance across drug classes in North America and Europe.
4. Krecek R.C. et al. (2018). Nigella sativa (black seed) against equine gastrointestinal parasites — Peer-reviewed trial showing significant FEC reduction in horses treated with black seed extract.
5. Stear M.J. et al. (2007). Host resistance to intestinal nematodes — mechanisms and genetics — Veterinary Immunology and Immunopathology — IgA, mast cells, and host immune mechanisms against gut parasites.
6. Proudman C.J. & Trees A.J. (1996). Tapeworms as a risk factor for equine colic — Equine Veterinary Journal — association between tapeworm burden and ileocecal colic in horses.
7. von Samson-Himmelstjerna G. (2012). Anthelmintic resistance in equine helminths — Veterinary Parasitology — mechanisms of resistance development and implications for management.
8. Chevalier G. et al. (2012). Earthing — health implications of reconnecting to Earth's electrons — J Environmental and Public Health — grounding, immune function, and oxidative stress reduction.
9. van Doorn D.C.K. et al. (2014). Practical aspects of pasture management to reduce parasite contamination — Equine Veterinary Education — rotational grazing, harrowing, and mixed species grazing evidence.
10. Nielsen M.K. (2016). Equine tapeworm infections — control, treatment and re-infection patterns — Equine Veterinary Journal — tapeworm biology, detection, and targeted treatment.
11. Bellaw J.L. et al. (2018). Anthelmintic therapy of equine ascarid infections — International Journal for Parasitology: Drugs and Drug Resistance — ivermectin resistance in ascarids and fenbendazole as alternative.
rootedsaviors.com | biofieldapp.com | stewardsunderpressure.com | Equine Wellness Blog



Comments